Structures and Dynamics of Hydrogen Cluster Materials Li5MoH11 and Li6NbH11
Yamamuro Group
In the current research field of batteries, there are strong demands for high-performance hydrogen storage materials and ionic (H+, Li+, etc.) conductors. Recently, a series of novel materials Li5MoH11, Li5WH11, Li6NbH11, Li6TaH11 which contain unusual ninefold hydrogen-coordinated clusters MH9 (M= Mo, W, Nb, Ta), whose structure is shown in the inset of Fig. 1, were synthesized by Orimo group in Tohoku University [1]. These are of interest since they have high hydrogen density and possible high Li ionic conductivity. The dynamics of MH9 cluster may play important roles in determining the physical properties of the materials. We have performed the neutron powder diffraction (NPD) and quasielastic neutron scattering (QENS) experiments of Li5MoH11 and Li6NbH11.
Both samples were synthesized from solid-state reactions under high pressure (ca. 5 GPa) and high temperature (ca. 700 K). Owing to these difficulties, the sample amounts of Li5MoH11 and Li6NbH11 were 33 mg and 20 mg, respectively. The NPD and QENS measurements were conducted using high intensity total diffractometer NOVA (BL21) and biomolecular dynamic spectrometer DNA (BL02) in MLF, J-PARC, respectively. The DNA data were collected with both high and low energy resolution modes.
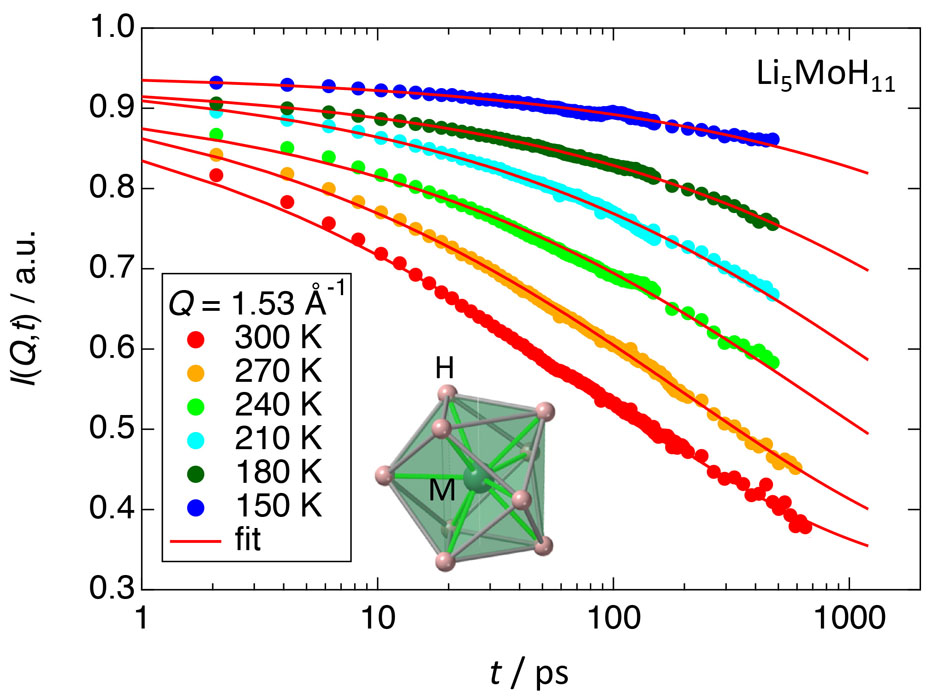
From the Rietveld analyses for the NPD data at room temperature, we have checked the purity of the samples (Li5MoH11: 75 %, Li6NbH11: 57 %) and confirmed that the MH9 clusters are orientationally disordered. Figure 1 shows the intermediate scattering functions of Li5MoH11. Similar data were obtained also in Li6NbH11. Relaxation phenomena were observed in a wide temperatures range between 150 K and 300 K. It is quite unusual that the relaxation occurs in a wide time range over 3 orders of magnitude as shown in the data at 300 K. Therefore, the data were fitted with not an exponential but Kohlraush-Williams-Watts (KWW) function
I(Q,t) = a(Q) + b(Q)exp[–(t/τKWW)β],
where Q is magnitude of a scattering vector, t is a time, τKWW is a relaxation time, β is a non-exponential parameter, and a(Q) and b(Q) are elastic and relaxation components, respectively. The β parameter (0 < β < 1) reflects the width of the relaxation time distribution; no distribution at β = 1. The fittings were satisfactory as shown by solid curves in Fig. 1. The determined β were 0.38 for Li5MoH11 and 0.33 for Li6NbH11, indicating that the distribution of the relaxation time is very wide; cf. β is around 0.5 for glass forming liquids. The activation energies obtained from the temperature dependence of the relaxation times were 15 kJmol–1 for Li5MoH11 and 27 kJmol–1 for Li6NbH11. The magnitude of the activation energy may depend on the ionic valence and size (M-H distance) of the hydrogen cluster (MoH93-: 1.76 Å, NbH94-: 1.86 Å) .
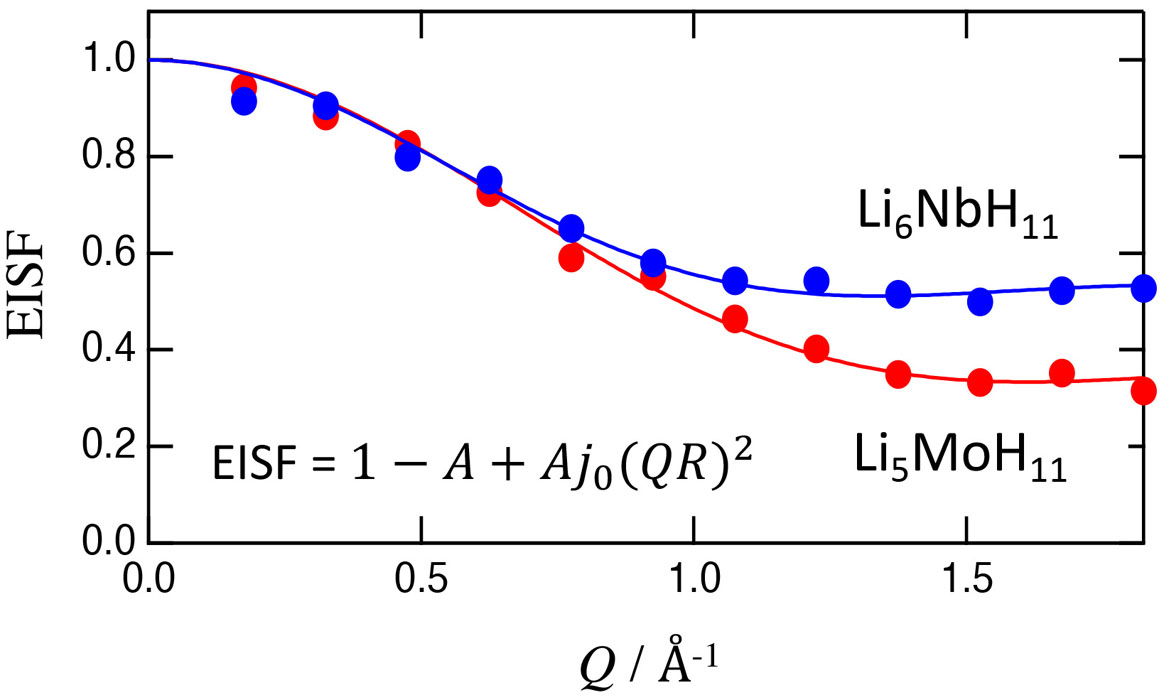
Figure 2 shows the Q dependence of EISF (Elastic Incoherent Scattering Factor) which is defined by EISF = a(Q)/[a(Q) + b(Q)]. As shown by the solid curves in Fig. 2, EISF was well fitted by
EISF = 1 – A + Aj02(QR),
where j0(QR) is a 0th order sphere Bessel function with a radius R. This indicates that MH9 clusters are spherically rotating at 300 K, which is consistent with the structural information. The rotational states of the MH9 clusters are now investigated with a first-principle MD simulation technique. We guess that the wide distribution of the relaxation time is associated with the positional disorder of Li ions conducting in the crystals.
This work is financially supported by JSPS KAKENHI No. JP 18H05513 and JP18H05518.
References
- [1] S. Takagi, Y. Iijima, T. Sato, H. Saitoh, K. Ikeda, T. Otomo, K. Miwa, T. Ikeshoji, and S. Orimo, Sci. Rep. 7, 44253 (2017).