Mass Transport in the PdCu Phase Structures during Hydrogen Adsorption and Absorption Studied by Ambient-Pressure XPS under Hydrogen Atmosphere
J. Tang, I. Matsuda, and J. Yoshinobu
Pd-based alloys have been the attractive materials for the hydrogen permeation process, because they possess higher hydrogen permeability and suppress the hydrogen embrittlement compared to pure Pd bulk. It was reported that the PdCu alloy, especially so-called “PdCu40 alloy”, has a relatively higher hydrogen diffusivity than the other Pd-alloys (PdAg, PdAu, PdRh, etc.). Industrial usages of the PdCu40 alloys with the high Cu content also improve the economic perspectives because of the lower Cu cost. Although there are advantages of PdCu alloys in the application fields, it requires further improvements in microscopic aspects. For example, hydrogen solubility of the face-centered cubic (fcc) structure of a PdCu alloy is approximately one order of magnitude higher than that of body-centered cubic (bcc) structure.
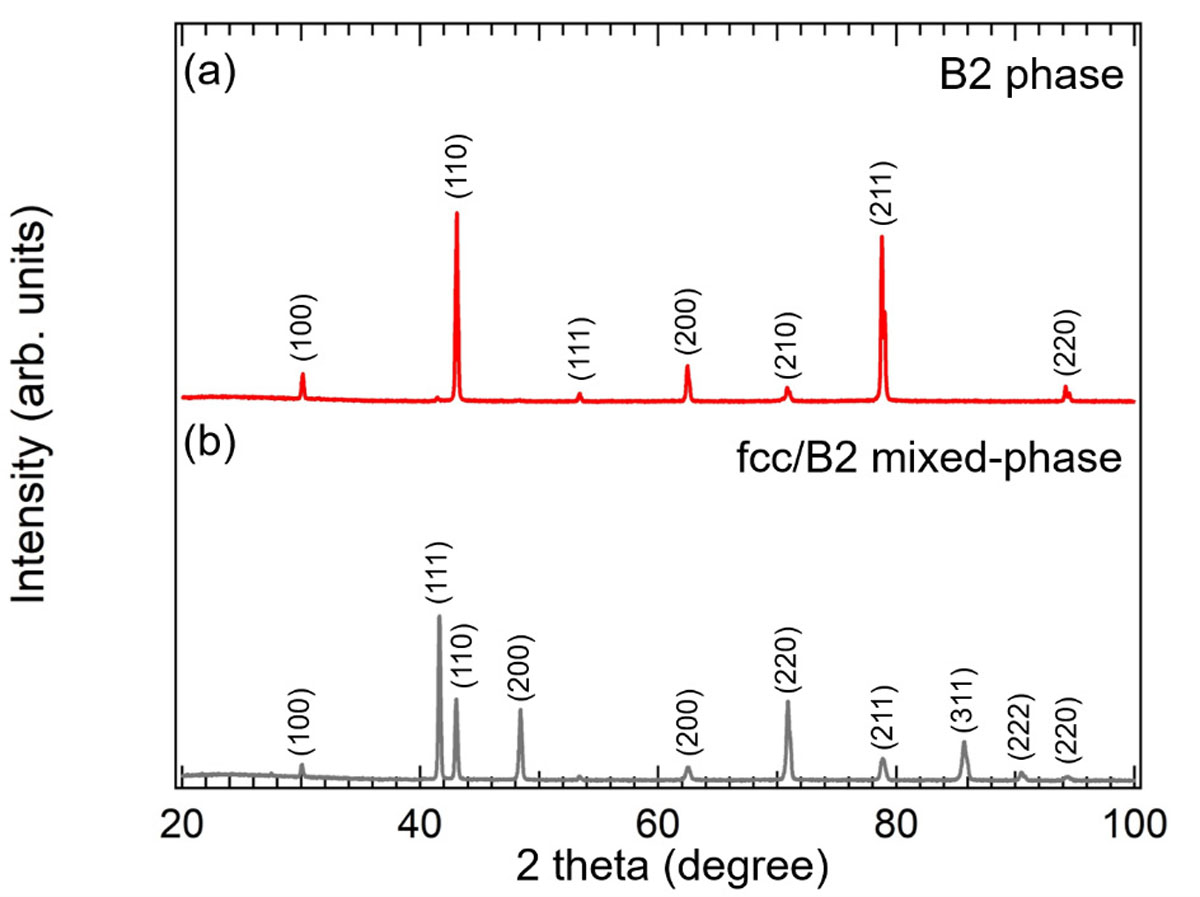
In the present research, we traced Pd and Cu atoms during hydrogen adsorption and absorption processes at the elemental steps, dissociative adsorption of H atoms on the surface and dissolution of H atoms into the bulk, by the individual core-level photoelectron spectroscopies under the hydrogen atmosphere [1]. The absorption process is investigated by the Ambient Pressure (AP)-XPS measurements at the soft X-ray undulator beamline BL07LSU at SPring-8. The PdCu40 sample, provided from Tanaka Kikinzoku Kogyo K.K., was annealed at 673 K to form the B2 phase structure. Then, the B2/fcc phase structure was prepared by subsequent annealing at 1000 K. Figures 1(a) and (b) show the XRD patterns of the B2- and fcc/bcc- phased PdCu alloys, respectively. Figures 2 and 3 show the temperature dependence of hydrogen absorption reactions on the B2 and fcc/bcc phases, observed by measuring the Pd 3d5/2 and Cu 2p3/2 XPS spectra during the hydrogen exposure at 1.5 Torr at 300-573K. The temperature-dependent data show that a number of the Pd-H bond increased accompanied with the decrease of the Cu-H bond with temperature. The Pd hydride formation in the B2 and fcc/bcc structures show the same behavior at 300-373 K, however, more Pd hydride was formed in the B2 structure than in the fcc/bcc structure at 473-573 K. The decrease rate of Cu-H component is higher in the B2 phase than fcc/bcc phase. These results indicate that the interstitial hydrogen occupied the different sites in the PdCu alloy bulk at different temperature and confirms the difference in absorption between the two phases. Comparing with the previous calculation [2], the significant enhancement of the hydride formation at 473 K can consistently be described in terms of variation of the hydrogen diffusion paths from the Cu-rich octahedral site to the energetically favorable Pd-rich octahedral site. Furthermore, the activation energy of the hydrogen diffusion is found to be lower in the B2 phase than that in the fcc/B2 phase. Our elucidation of hydrogen diffusion mechanisms on the different phase structures gives a new understanding of the hydrogen absorption process, which could be the critical point for the rate-limiting process in the hydrogen permeation through the PdCu alloy.
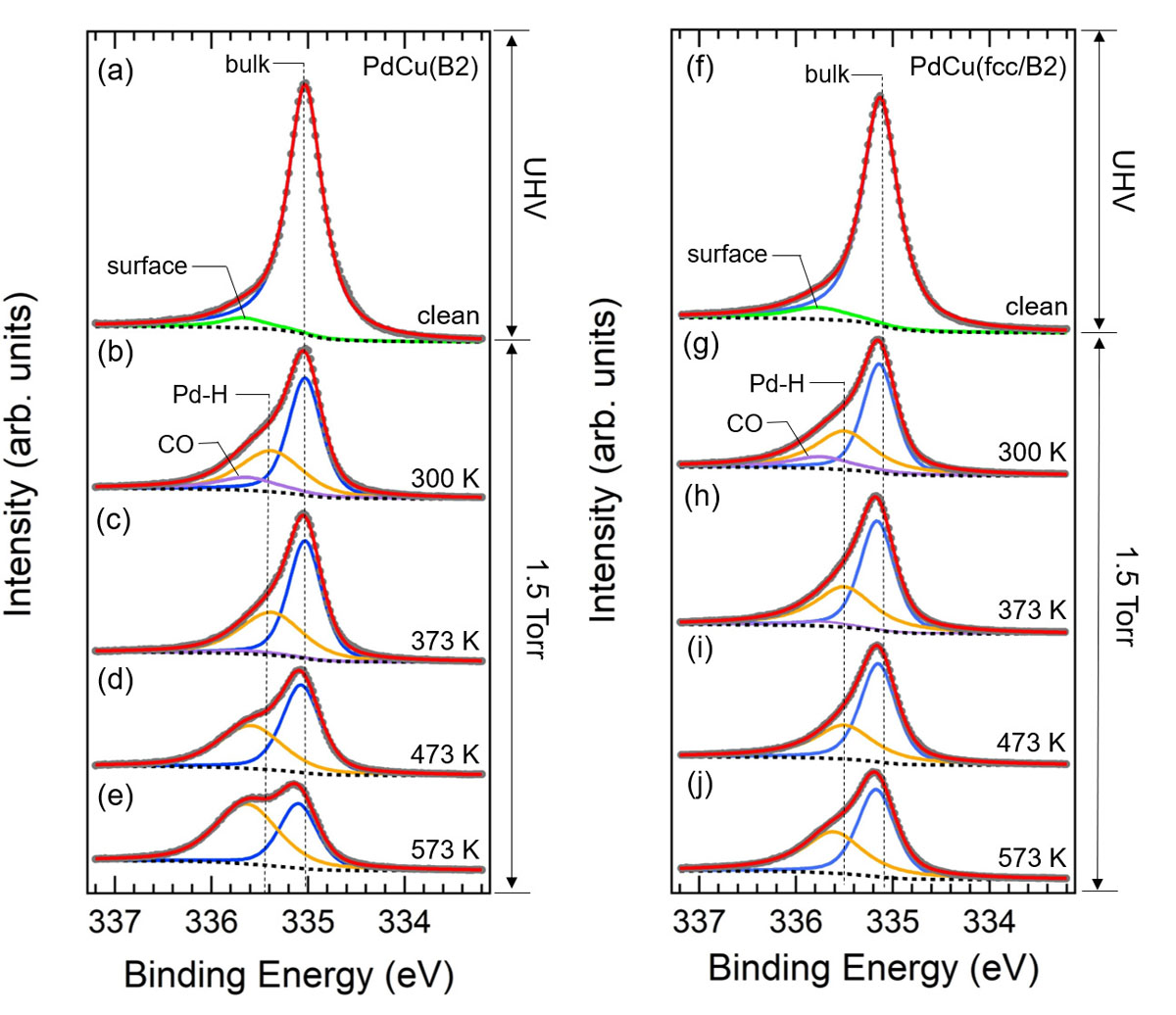
Fig. 2. Pd 3d5/2 spectra measured on (a) clean surface, and under H2 exposure at 1.5 Torr at (b) 300 K, (c) 373 K, (d) 473 K, (e) 573 K with photon energy of 680 eV on PdCu(B2) alloy. Pd 3d5/2 spectra measured on (f) clean surface, and under H2 exposure at 1.5 Torr at (g) 300 K, (h) 373 K, (i) 473 K, (j) 573 K with photon energy of 680 eV on PdCu(fcc/B2) alloy. Surface Pd component, bulk Pd component, CO adsorption component and Pd-H component are presented with green, blue, purple, and yellow solid lines, respectively.
Similar investigation on hydrogen permeation process was carried out for the PdAg alloy[3]. The measurements of AP-XPS revealed that Pd atoms were also found to reactive sites during hydrogen absorption. On the other hand, the process is likely promoted by the Pb segregation. It is of interest to make systematic investigations of individual Pd-alloys to capture the whole picture.
References
- [1] J. Tang, S. Yamamoto, T. Koitaya, A. Yoshigoe, T. Tokunaga, K. Mukai, I. Matsuda, and J. Yoshinobu, Appl. Surf. Sci. 480, 419 (2019).
- [2] L. C. Liu, J. W. Wang, Y. H. He, and H. R. Gong, J. Membr. Sci. 542 , 24 (2017).
- [3] J. Tang, S. Yamamoto, T. Koitaya, Y. Yoshikura, K. Mukai, S. Yoshimoto, I. Matsuda, and J. Yoshinobu, Appl. Surf. Sci. 463, 1161 (2019) .