Photoinduced Hydrogen Release from Hydrogen Boride Sheets
I. Matsuda Group
Materials consisting of one atomic layer, such as graphene, are one of the two-dimensional materials that have shown various physical and chemical properties that are different from ordinary ones. Recently, novel atomic layers of boron, so-called “borophene”, were reported and the materials were found to surpass the performance of graphene by theoretical works. However, the growth requires special conditions such as appropriate metal substrates, high temperature and ultra-high vacuum.
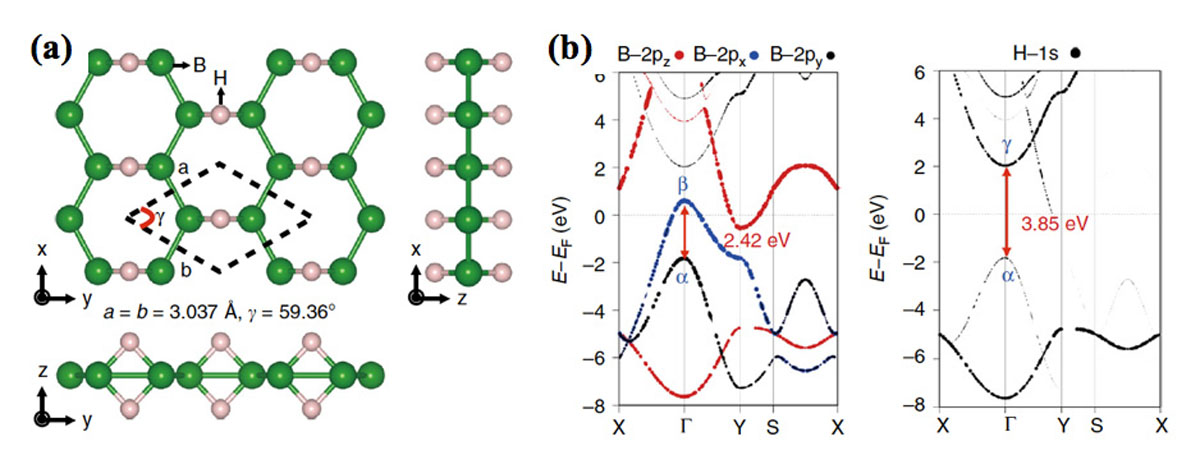
Under these circumstances, we have succeeded in synthesizing a hydrogen boride (HB) sheet (Fig. 1(a)), which is hydrogenated borophene or borophane, under the ambient condition. Since the HB sheet is composed only of the light elements, boron and hydrogen, the mass hydrogen density is as high as 8.5%, and it was expected to be applied as a safe and lightweight material for the hydrogen carrier that replaces the conventional high-pressure hydrogen gas cylinder with a risk of explosion. However, it has required to consider a procedure to extract hydrogen atoms/molecules from the sheet before the actual use. In the present research, we designed and verified the hydrogen release by the light irradiation [1].
According to the first-principles calculation of the hydrogen boride sheet, the optical transition (α → β) occurs from the bonding orbital of boron to the antibonding orbital, as shown in Fig. 1(b). On the other hand, there is also an optical transition (α → γ) to the antibonding orbital of hydrogen. Since this transition energy corresponds to a photon energy of ultraviolet (UV) ray, it is hypothesized that hydrogen could be released from a hydrogen boride sheet only by the UV irradiation even under the ambient condition.
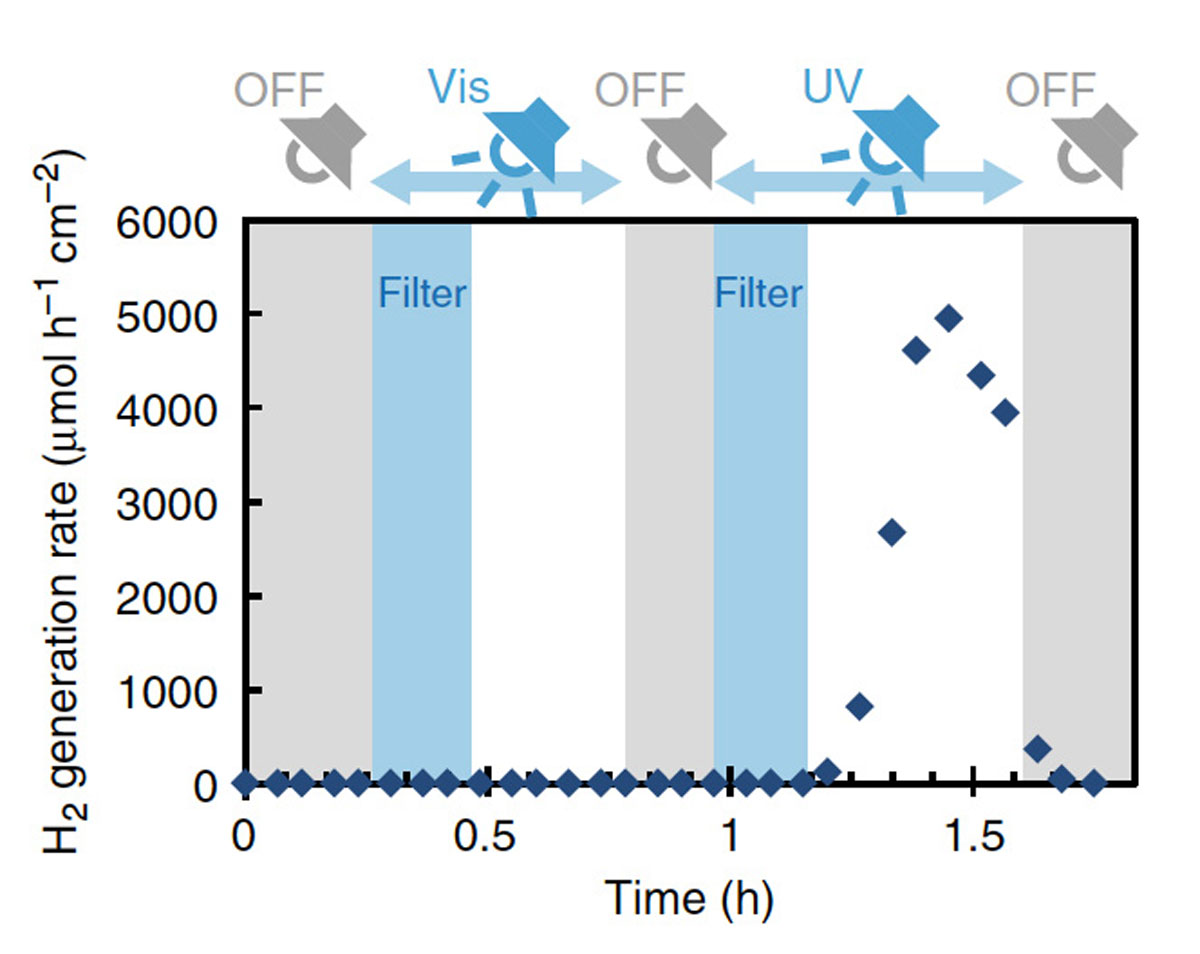
The experimental verification was made by analyzing the gas molecules emitted from the hydrogen boride sheets with using two types of light sources. One type of the sources irradiates visible light that can cause the optical transition (α → γ), while the other type irradiates ultraviolet light that can induce the optical transition (α → β). As shown in Fig. 2, it was confirmed that the hydrogen gas was generated by exposure of the ultraviolet ray. By quantifying the amount of the hydrogen production, it was found that 8% of the mass of the hydrogen boride sheet was consumed for the hydrogen release. For conventional hydrogen storage alloys, the mass hydrogen density is up to 2%. Even a promising hydrogen carrier of the organic hydrides reaches mass hydrogen density of 6.2% with heating above 300 °C. Thus, the hydrogen boride sheet can be a safe, lightweight and simple portable hydrogen carrier with a simple light operation of the extremely high hydrogen release.
References
- [1] R. Kawamura, N. T. Cuong, T. Fujita, R. Ishibiki, T. Hirabayashi, A. Yamaguchi, I. Matsuda, S. Okada, T. Kondo, and M. Miyauchi, Nature Commun. 10, 4880 (2019).