Ruddlesden-Popper Quantum Wells
Lippmaa Group
Several different approaches have been proposed for fabricating two-dimensional oxide quantum wells. In most cases, the techniques known from traditional semiconductors have been applied to oxides. However, epitaxial oxide heterojunctions or superlattices do not generally show good carrier confinement. One of the main reasons for this is the complicated structural and electronic distortions that occur at oxide heterointerfaces. Charge transfer across an interface and the formation of very thick depletion layers mean that quantum well structures that offer excellent structural confinement do not generally provide good electronic confinement.
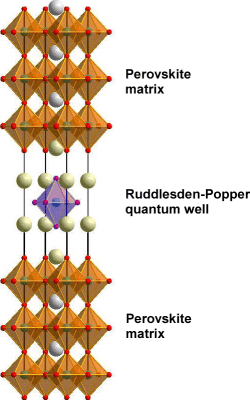
Fig.1. Structure of a quantum well formed by embedding a single Ruddlesden-Popper formula unit layer in a perovskite matrix.
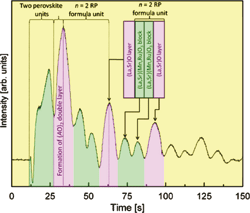
Fig. 2. Specular RHEED intensity during the growth of a La1.2Sr1.8Mn1.7Ru0.3O7 film. The alternating sequence of rocksalt (La,Sr)O and perovskite (La,Sr)(Ru,Mn)O7 blocks can be seen. In this case, the growth started with the formation of a double perovskite block at the interface.
We are attempting to solve this problem by fabricating a quantum well that combines a cubic perovskite lattice with a Ruddlesden-Popper (RP) phase. This combination is similar to a bulk RP material with the exception that we embed only a single RP formula unit in a perovskite matrix, as illustrated in Fig. 1.
This structure has several advantages over a simple perovskite heterojunction. The center part of the well structure has the same cation coordination and lattice parameter as the matrix material, but it is isolated from the matrix by rocksalt layers on both sides. The rocksalt layers provide both electronic confinement and ionic confinement, i.e. the structure can be thermodynamically stable and thus resistant to cation interdiffusion.
An essential part of growing such structures is the ability to monitor the growth of each atomic layer. We do this by measuring the intensity of the specular reflection in a reflection high-energy electron diffraction pattern. The intensity variation with time (Fig. 2) shows that we can indeed observe the formation of each perovskite and rocksalt block. In this example, a La 1.2Sr1.8Mn1.7Ru0.3O7 film was grown on a SrTiO3 surface. By suitable buffering of SrTiO3 substrate surface with a fractional layer of (La,Sr)O, it is possible to choose which block forms at the initial interface, either a perovskite unit or a rocksalt unit.
References
- M. Matvejeff, T. Chikyow, and M. Lippmaa, Journal of Crystal Growth 311, 1201 (2009).
